Research
has consistently shown that people with HIV/AIDS have an elevated risk for many
types of cancer, especially those caused by an infectious pathogen such as human
papillomavirus (HPV), which can cause cervical, anal, genital, and oral malignancies.
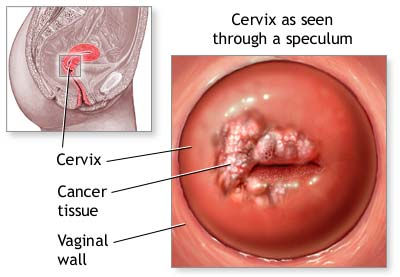 |
The
Pap smear is both a visual inspection and a culture of the cervix. An instrument
called a speculum holds the walls of the vagina open so that the cervix may be
viewed and a swab of cells obtained for culture and analysis. |
|
Current
HIV treatment guidelines recommend that HIV positive women should received
annual Papanicolaou (Pap) tests to detect precancerous cell changes or early stages
of cancer (dysplasia or neoplasia). Annual tests used to be recommended for HIV
negative women as well, but women with repeated normal tests and no risk factors
are now advised to get a Pap smear every 2-3 years.
Alexandra
Oster from the U.S. Centers for Disease Control and Prevention (CDC) and colleagues
assessed screening prevalence and associated factors among HIV positive women
in 18 states. The analysis was based on data collected in interviews of people
with HIV conducted during 2000-2004.
Results
 | Out
of 2417 women surveyed, 556 (23.0%) did not report receiving a Pap test during
the past year.
|
 | Compared
with those who had a recent test, women without a recent Pap test were more likely: |
 | to
be older (adjusted odds ratio [OR] 1.3 per 10 years); |  | to
have their most recent CD4 count < 200 cells/mm3 (adjusted OR 1.6); |  | to
have an unknown CD4 cell count (adjusted OR 1.4). |
|
 | The
likelihood of a missed Pap test increased among women who did not received their
most recent pelvic exam from their usual source of HIV care (adjusted OR 2.6).
|
"HIV
care providers should ensure that HIV-infected women receive annual Pap tests,
recognizing that missed Pap tests are more likely among older women and women
with low CD4 cell counts," the study authors concluded. "Integrating
HIV and gynecologic care and educating clinicians about recommendations may increase
screening."Epidemic
Intelligence Service, Office of Workforce and Career Development, Centers for
Disease Control and Prevention, Atlanta, GA.
8/14/09
Reference
AM
Oster, PS Sullivan, and JM Blair. Prevalence of cervical cancer screening of HIV-infected
women in the United States. Journal of Acquired Immune Deficiency Syndromes
51(4): 430-436. August 1, 2009. (Abstract).